
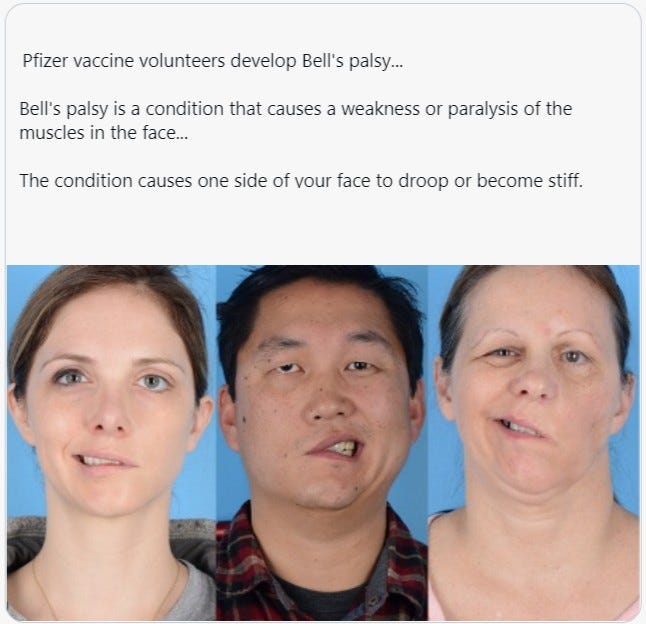
Is that Bell's Palsy, a stroke, or did you just have a Pfizer vaccine?
OK. Vaccine injuries are definitely not funny. But it is unbelievable that people still took these vaccines after hearing about the initial side effects that people were encountering early on. In the beginning, it's understandable why people were swayed. There was an extensive propaganda effort to ensure widespread vaccination, leading to multiple doses being administered—now, who even knows how many shots we're on??


Similar to Moderna, Pfizer was eager to bring their vaccine to market at WARP SPEED in response to the early stages of the pandemic. Led by CEO Albert Bourla, Pfizer embarked on an ambitious development and distribution strategy to combat the global health crisis. Bourla, a seasoned executive with a background in veterinary medicine, has been instrumental in steering Pfizer's efforts towards vaccine development and deployment. Under his leadership, Pfizer collaborated closely with BioNTech to develop their mRNA-based COVID-19 vaccine, which eventually received emergency use authorization from regulatory authorities around the world. Bourla's leadership and Pfizer's rapid response played a crucial role in delivering a vaccine that significantly contributed to the fight against the pandemic.
How was Pfizer able to distribute their vaccines so rapidly, almost as if it were planned ahead of time?
Corning Incorporated, an established technology company with over 170 years of history, entered the realm of vaccine vial production during the mid-20th century, coinciding with the development of the first polio vaccine by Dr. Jonas Salk. The polio vaccine vials manufactured by Corning were produced around seven decades ago. Corning's proficiency in glass technology enabled the development of vials suitable for storing and transporting the vaccine. However, the timing of their involvement raises suspicions, particularly given that Dr. Jonas Salk's research was supported by The March of Dimes. The organization, initially named the National Foundation for Infantile Paralysis, founded by President Franklin D. Roosevelt in 1938 purportedly to address polio, despite Roosevelt's associations with eugenics.
The Rockefeller family's participation in these matters also invites scrutiny, considering John Rockefeller's advocacy for eugenics and his significant involvement in the purported polio epidemic response of that era.

Could Corning's sudden foray into vaccine vial production simply be driven by opportunism, given the lucrative nature of the pharmaceutical industry? Is there a possibility that vaccine vials could be utilized for malicious purposes, perhaps containing undisclosed toxic coatings that induce illness? It raises concerns about the extent to which we can trust institutions like the FDA to thoroughly oversee and guarantee safety, especially considering past instances of dubious practices. This uncertainty highlights the challenges in ensuring transparency and accountability within regulatory frameworks.
Corning Incorporated began developing Corning Valor® Glass technology, the novel vial technology utilized for the Pfizer COVID-19 vaccines, several years prior to its commercial collaborations with pharmaceutical giants such as Merck and Pfizer. The inception of Corning Valor® Glass can be traced back to the mid-2010s, although the precise start date of its development within Corning remains undisclosed as does the exact propriatary compostion of the vials themselves.

In efforts purported to enhance pharmaceutical packaging standards and increase production capacities, both Merck & Co., Inc. and Pfizer collaborated with Corning Incorporated in 2017 to integrate Corning Valor® Glass vials into their vaccine and biologics packaging.
In November 2017, Corning Inc. announced plans to manufacture Valor Glass in the Southern Tier, creating 185 new jobs with support from New York state, including over $7 million in funding. Governor Andrew Cuomo visited Corning to unveil the state's financial assistance, emphasizing the importance of job creation and economic growth. Corning Valor® Glass, developed at Corning's Sullivan Park Research Facility was a project totaling $95.5 million. It generated employment at the Big Flats Innovation Support Center and Sullivan Park.
Cuomo's association with this project raises some suspicions, particularly due to its timing, which preceded the COVID-19 pandemic in what appears to be an oddly fortuitous manner. His involvement prompts inquiries into Cuomo's support for the project and the potential presence of conflicts of interest or preconceived planning. These suspicions are amplified by concerns surrounding transparency and accountability, particularly evident during Cuomo's administration's handling of the COVID-19 pandemic response. The administration faced criticism, including allegations of underreporting nursing home deaths, policies that increased nursing home fatalities, and mismanagement of resources, which notably marred Cuomo's leadership during the crisis.
In 2018, during an event held in Durham, North Carolina, Corning fortuitously unveiled plans for another new facility dedicated to manufacturing Corning Valor® Glass. It's remarkable how all of this groundwork was laid before the pandemic, with help from Pfizer, Merck and governmental institutions, just in time for this novel products critical demand.
The attendance at the launch of the new factory for manufacturing Corning Valor® Glass in 2018 was remarkable. Yet, one may ponder the hastiness of its opening. Why the big rush? Between 2014-2015, there were purported measles outbreaks supposedly traced back to an individual from Disneyland in California and other claimed instances in New York in 2018. But those supposed outbreaks were contained. Curiously, these outbreaks served as the catalyst for the removal of religious exemptions for vaccines in these states, conveniently preceding the COVID-19 pandemic( a topic deserving of thorough examination).
Additionally, there were reports of localized outbreaks of the purported Ebola virus in the Congo and Zika virus in Brazil, but these outbreaks remained contained within specific regions.

So what was the big rush in 2018 to get these novel vials made? Unlicensed Dr. Gates who never completed college, would you like to take the floor?
It's intriguing that in October 2019, Bill Gates and his associates organized what appears to be a simulated viral outbreak scenario, just a month before the purported first case of COVID-19 emerged…
During October 2019, while Event 201 was taking place, Corning Incorporated revealed that a prominent pharmaceutical manufacturer had obtained FDA approval for Corning Valor® Glass to be utilized as a new primary package for a marketed drug product. However, Corning did not disclose the identity of this pharmaceutical company. This secrecy appears peculiar, prompting questions about the reasons behind it. And the question arises: why?
This novel technology was announced in 2017 and seems to have been around for years prior to this. So why wasn’t it being utilized? Merck and Pfizer team up with Corning to innovate pharmaceutical glass packaging, introducing Corning Valor™ Glass on July 20, 2017.

This advanced solution improves drug storage and delivery, endorsed by the White House for its collaborative and economic impact. President Trump commends the initiative, highlighting a $500 million investment by Corning, leading to 1,000 new jobs, with plans for a total $4 billion investment and 4,000 jobs. Merck's CEO emphasizes the potential of Valor Glass to revolutionize pharmaceutical packaging.
In July 2017, at “Made in America,” week, a special White House event celebrating American manufacturing, President Trump did his best to break a new Valor vial. The vial showed its unique formulation and strength. It remained intact even after he exerted 1,000 pounds of force — the equivalent of a punch from a professional boxer.
Why was this supposedly novel vial granted FDA approval in October 2019 mere weeks before the supposed COVID-19 pandemic commenced?

On June 09, 2020, Corning Incorporated, in collaboration with the U.S. Departments of Defense (BARDA) and Health & Human Services, received $204 million in funding under the Operation Warp Speed Initiative. This funding aimed to expand domestic manufacturing capacity for Valor® Glass packaging for COVID-19 vaccines and treatments.

It is interesting to note that Pfizer signed a long-term purchase agreement with Corning for their supposedly novel vials for COVID-19 vaccine manufacturing (and even though they had already announced this collaboration back in 2017).

The link above that states granted also comes back with a…
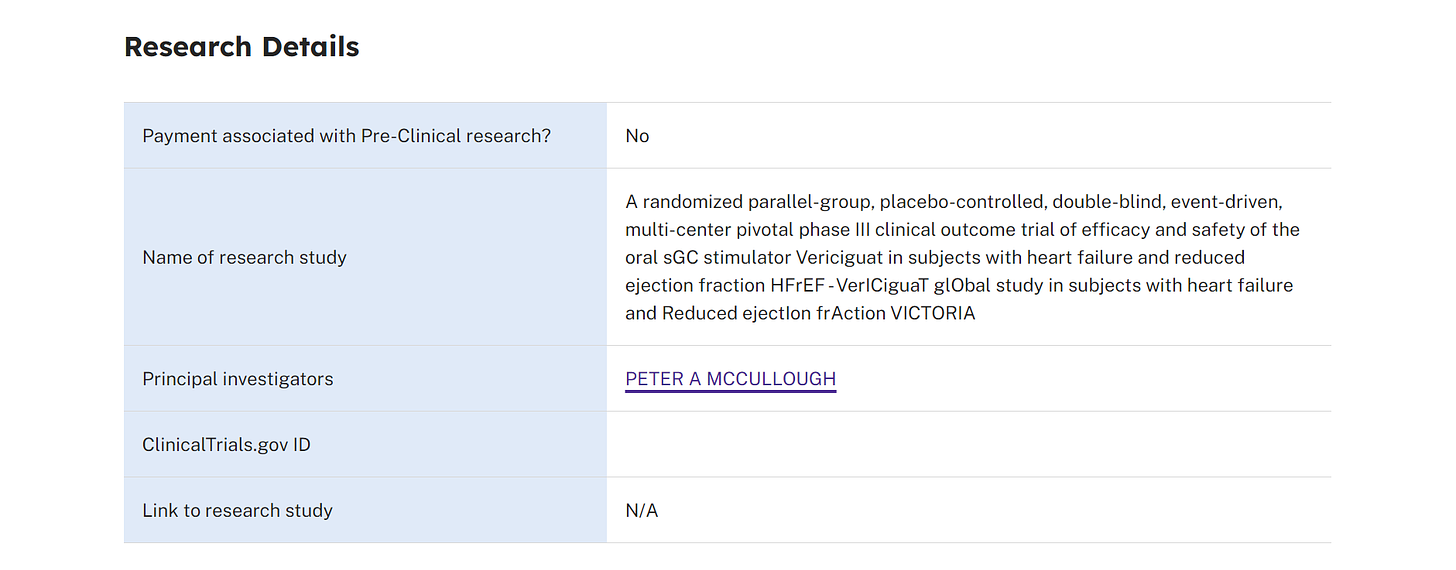
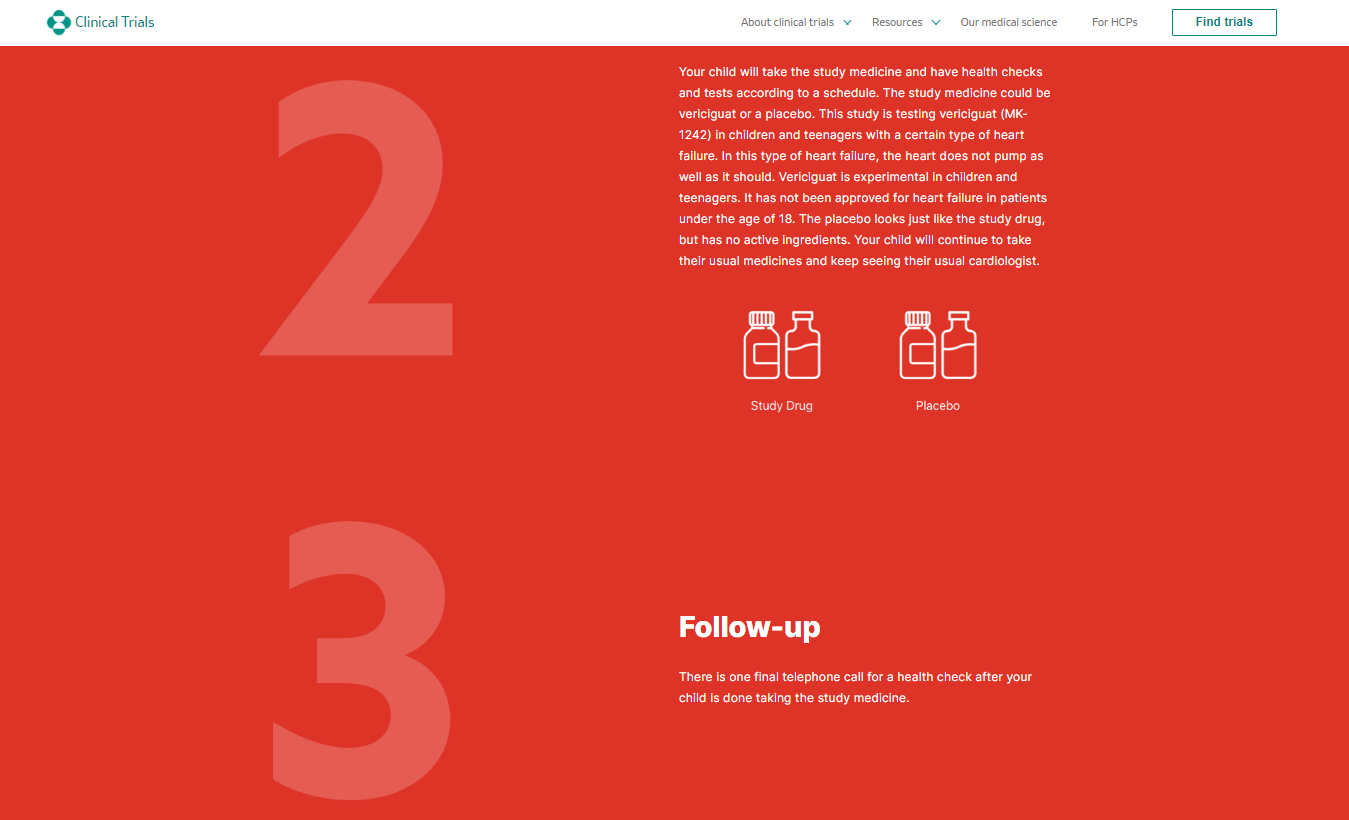
In yet another peculiar set of coincidences, on January 20, 2021, Merck (who had also partnered with Corning for the novel glass vials in 2017) received FDA approval for a novel medicationVerquvo (vericiguat), a soluble guanylate cyclase (sGC) stimulator, to reduce the risk of cardiovascular death and heart failure hospitalization in adults with symptomatic chronic heart failure and ejection fraction less than 45%. The timing is opportune for introducing a medication designed for these particular cardiac conditions, especially considering that individuals who have received COVID-19 vaccines are experiencing similar cardiac issues.
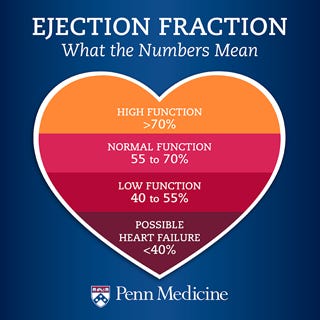
Developed jointly with Bayer AG, Verquvo is the first treatment specifically approved for patients following a heart failure hospitalization or need for outpatient IV diuretics. The approval, based on the VICTORIA trial results, marked a milestone in cardiovascular medicine. Verquvo 's efficacy was demonstrated in reducing the risk of cardiovascular death or heart failure hospitalization compared to placebo. What timing!
Verquvo, a novel treatment for heart failure, sparks skepticism due to its introduction by Merck amidst reports of heart-related issues associated with certain COVID-19 vaccines. Despite collaboration with Bayer to supposedly address medical gaps, concerns also arise regarding Verquvo's safety, notably highlighted by its boxed warning against use in pregnant females due to potential fetal harm. This caution, coupled with Merck's timing and ongoing concerns regarding vaccine-related heart complications, highlights the serious need for thorough scrutiny and consideration of potential risks.
It seems that Dr. Peter A. McCullough, a well-known cardiologist, may have been involved as a principal investigator in this drug trial, as indicated by CMS Open Payments. However, details regarding his exact role in the trial are elusive and difficult to ascertain beyond this information.
What's particularly noteworthy is the involvement of individuals positioning themselves as advocates for health freedom, purportedly aiming to establish alternative healthcare systems, are involved in drug trials such as the Victoria drug trial sponsored by pharmaceutical companies like Merck Sharp & Dohme LLC.
Merck Sharp & Dohme (MSD) LLC, known as Merck & Co. in the United States and Canada, is a multinational pharmaceutical company. It is one of the largest pharmaceutical companies in the world, involved in the research, development, manufacture, and distribution of a wide range of pharmaceutical products for human and animal health.
Merck Sharp & Dohme focuses on various therapeutic areas, including oncology, vaccines, infectious diseases, cardiovascular diseases, diabetes, and more. The company has a long history dating back to the 19th century and has a significant global presence.
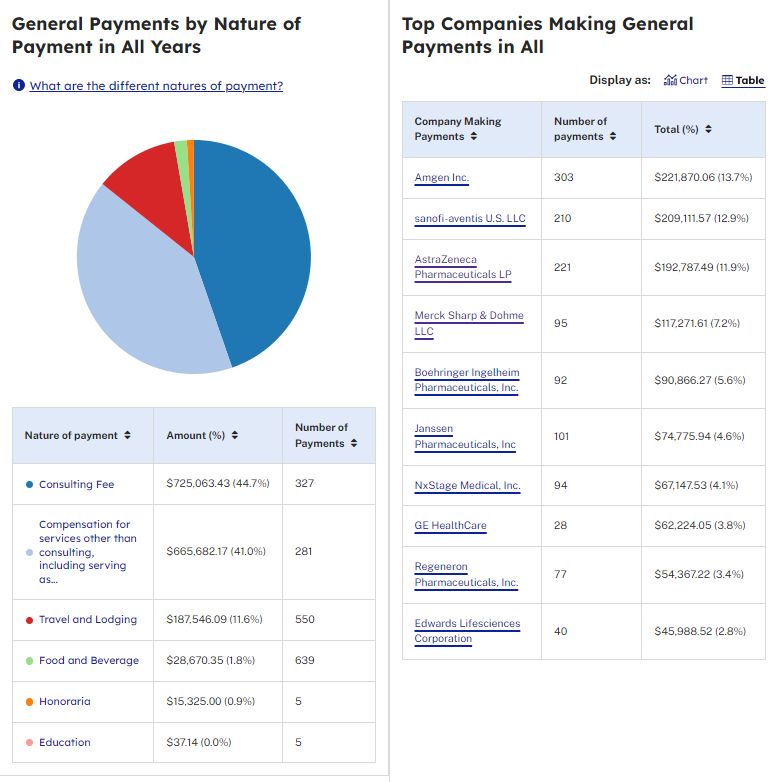
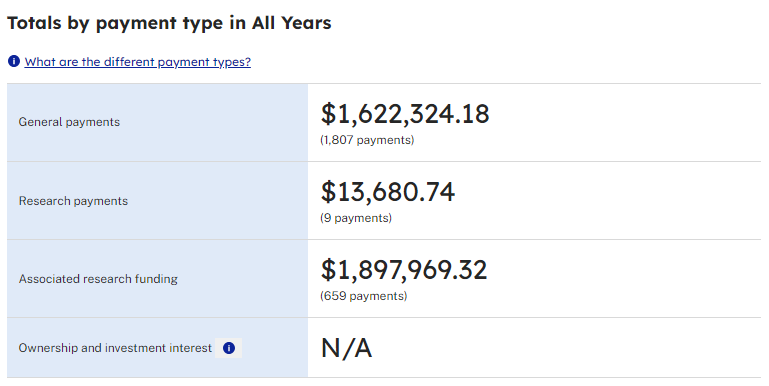
According to CMS Open Payments data spanning from 2016 to 2022, it appears that cardiologist, Dr. Peter McCullough, who over the past few years presents himself as being opposed to large pharmaceutical companies, has received $117,271.61 in general payments solely from Merck Sharp & Dohme LLC during this period. As of the writing of this Substack, the data for CMS Open Payments for the year 2023 is unavailable. It will be interesting to see what Dr. Peter McCullough received from pharmaceutical companies during 2023 when the information is released.
From 2016 to 2022, Dr. Peter McCullough received the following amounts from drug and/or medical device companies, as reported by CMS Open Payments. The arrangement between pharmaceutical and medical device companies appears to present a significant conflict of interest. When physicians and researchers are compensated to participate in clinical trials or research, it raises doubts about the impartiality of the data. This situation seems highly dubious and calls into question the trustworthiness of such data to the public.
How the majority of pharmaceuticals make it past clinical trials, receives FDA approval, and is given to the unwitting public who buy that these substances have been thoroughly tested and are safe is baffling!
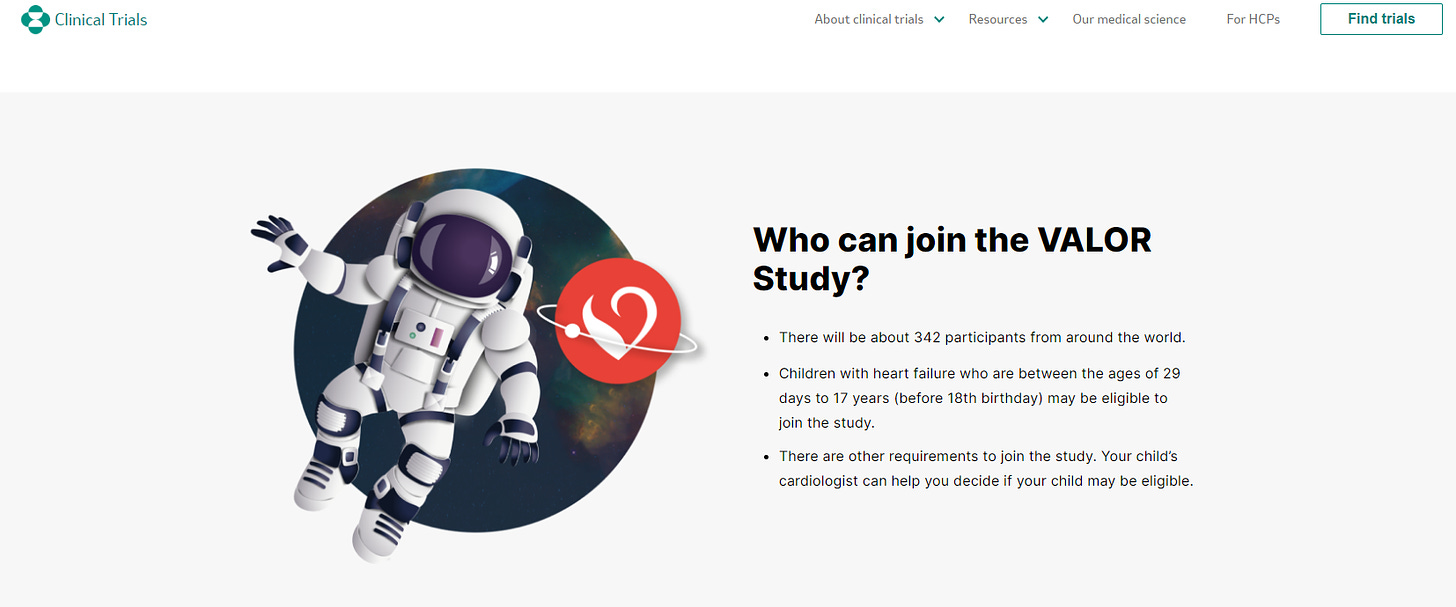
What's particularly concerning is Merck's choice to title the clinical trial for their heart failure medication, Verquvo, aimed at infants, children, and teenagers as the "Valor HF" study. Interestingly, "Valor" is the same name given to the innovative Corning vials specifically manufactured for Pfizer vaccines.
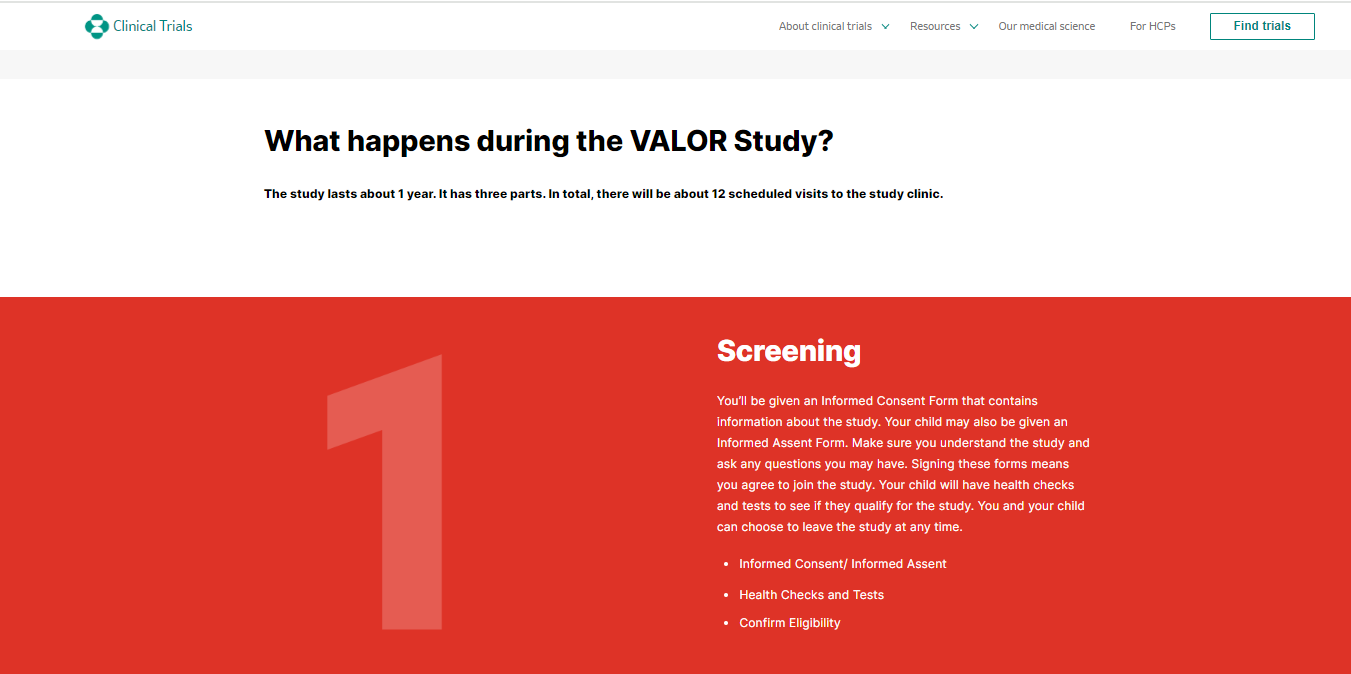
The ongoing Merck VALOR Study, partnered with Bayer, initiated May 31, 2023 and is estimated to be completed on April 30, 2030. The study seeks to evaluate the safety and potential efficacy of an experimental medication for infants, children, and teenagers with heart failure. Again, what impeccable timing for this medication in this specific population after the rollout of the Pfizer vaccines in this population!
Valor clinical trials refers to a series of research studies conducted to evaluate the safety and effectiveness of investigational drugs or treatments. These trials typically involve testing the medication on human subjects to assess its potential benefits and any associated risks. The goal of valor clinical trials is to gather scientific evidence that can inform healthcare decisions and ultimately improve patient outcomes. There are numerous clinical valor trials that have been conducted in the past and that are still ongoing.
What a truly interesting name for Corning to have named their novel vaccine vials that are now being utilized for Pfizer COVID-19 vaccines! Does this this mean that anyone who has received these Valor contained shots are in an ongoing clinical drug trial? Kinda seems that way! And much like the Moderna COVID-19 vials specially made by SiO2, I would like to know what is really on the inside of the Corning Valor® Glass vials, how is it interacting with the contents of the Pfizer “vaccine”, and what is it really doing to people who get these injections? Why were these Valor vials announced in 2017, manufacured in several factories for years, but were not utilized until supposedly 2021? Someone really needs to look into that.














Love your work and depth of exposure. It all points to LuciPfizer…
https://peter70x7.substack.com/p/lucipfizers-venomous-turbo-cancer
…and his fallen angels…
https://peter70x7.substack.com/p/last-call-to-repent-from-ishtar-sunday
Great documentation. I appreciate your focus on the glass used to store the vaccines. If you dig into the literature on aluminum contamination of intravenous medications you will find that the glass in which the medications are stored are the primary source of aluminum. I suspect that a lot of proprietary glass formulations are developed and marketed on their ability to reduce aluminum contamination. Polymer coating the glass is one way to achieve this, which is possibly one of the special things about the Corning Valor glass. A polymer film on the inside of the vial would potentially open up avenues for adulteration.